AN expert panel on Tuesday (12) recommended the use of Covaxin, one of the two major anti-coronavirus vaccines used in India and made by Bharat Biotech, on children aged between two and 18.
The company, which is based in Hyderabad, said, “Bharat Biotech has submitted data from clinical trials in the two - 18 age group for Covaxin to CDSCO (Central Drugs Standard Control Organisation). The data has been thoroughly reviewed by the Subject Expert Committee (SEC)... provided positive recommendations.”
ALSO READ: Rich nations to allow 100m Covid vaccines go waste
“This represents one of the first approvals worldwide for COVID-19 vaccines for the two - 18 age group... We now await further regulatory approvals prior to product launch and market availability of Covaxin for children,” it added.
Sputnik V jab gets a blow as India’s private vaccine sales slump
Covaxin is currently being used on adults in India’s ongoing Covid-19 vaccination drive. It is the second Covid-19 jab to get approved for children in India. The Drugs Controller General of India (DGCI) had in August approved ZyCoV-D for children aged 12 and above besides adults. The rollout is yet to begin. A third potential vaccine for children is Serum Institute’s Novavax for which the DGCI recently cleared trials for children between seven and 11 years. A fourth is Biological E’s Corbevax for kids above five.
India’s minister of state for health Dr Bharti Pravin Pawar told Indian news channel NDTV, “We (always) said we will get a vaccine for children between two and 18... it will be approved soon. SEC has recommended... it will be approved soon... trials were conducted keeping in mind safety of children. We are committed to their safety.”
The final approval will be given by the DGCI.
Last week, Bharat Biotech said it submitted data on vaccine trials on children.
The Covaxin vaccine tested on children is the same formulation which is used on adults. But separate trials were required to guarantee safety and efficacy on minors.
While data on the trials on children have not been made public yet, tests were carried out on 1,000-plus children across the country. The expert panel also said that the trial on kids showed similar efficacy rates as that on adults.
India is gradually turning its focus towards inoculating children against the virus, having administered nearly 96 crore doses to adults.
Dr Randeep Guleria, chief of Delhi’s All India Institute of Medical Sciences, has said that children in the two-18 age group must be vaccinated “because that's the only way to get rid of the pandemic”.
In early October, Dr NK Arora, the chief of India’s vaccine task force, told news agency Asian News International that children with severe comorbidities would be given priority while other (healthier) kids would be immunised subsequently.
“We are trying to identify (children) at highest risk... within the next couple of weeks, the list will be in the public domain. We are also making arrangements so these children do not have to travel (too far) to get the vaccine…,” he said.
India is the middle of resumption of schools and with teaching and non-teaching staff members returning to classrooms, concerns have been expressed over children getting infected besides the need to vaccinate adults working at schools.





 BJP supporters celebrate as the party takes lead during the West Bengal Assembly elections result day, at Raiganj, in Uttar Dinajpur district, Monday, May 4, 2026.PTI
BJP supporters celebrate as the party takes lead during the West Bengal Assembly elections result day, at Raiganj, in Uttar Dinajpur district, Monday, May 4, 2026.PTI In this screengrab from a video posted on May 4, 2026, Family members of Tamilaga Vettri Kazhagam (TVK) chief Vijay, including S A Chandrasekhar and Shoba, celebrate as early trends show the party leading in the Tamil Nadu Assembly elections, in Chennai. PTI
In this screengrab from a video posted on May 4, 2026, Family members of Tamilaga Vettri Kazhagam (TVK) chief Vijay, including S A Chandrasekhar and Shoba, celebrate as early trends show the party leading in the Tamil Nadu Assembly elections, in Chennai. PTI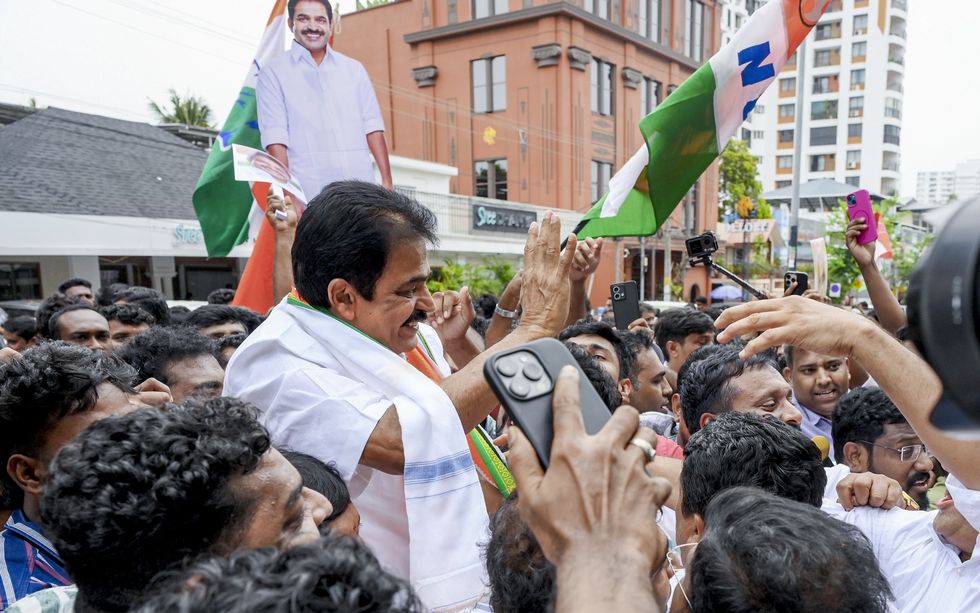 Congress MP KC Venugopal celebrates with party workers as the party takes lead during the Kerala Assembly elections result day outside KPCC office, in Thiruvananthapuram, Monday, May 4, 2026. PTI
Congress MP KC Venugopal celebrates with party workers as the party takes lead during the Kerala Assembly elections result day outside KPCC office, in Thiruvananthapuram, Monday, May 4, 2026. PTI Artists play traditional drums as BJP workers celebrate the party taking lead during the Assam Assembly elections results day, at party headquarters, in Guwahati, Monday, May 4, 2026. PTI
Artists play traditional drums as BJP workers celebrate the party taking lead during the Assam Assembly elections results day, at party headquarters, in Guwahati, Monday, May 4, 2026. PTI Puducherry: Poll officials during the counting of votes for the Puducherry Assembly elections at a counting centre, in Puducherry, Monday, May 4, 2026. PTI
Puducherry: Poll officials during the counting of votes for the Puducherry Assembly elections at a counting centre, in Puducherry, Monday, May 4, 2026. PTI







